Electron pair geometry chart of asf33/10/2024 AsF 3 Lewis Structure AngleĪngle denotes basically the angle between two bonds. Formal charge of fluorine (F) = 7 – 6 – (2/2) = 0įrom the formal charge calculation, it is clear that AsF 3 is a totally neutral molecule with zero charge.Formal charge of arsenic (As): 5 – 2 – (6/2) = 0.Formal charge = Total number of valance electrons – number of electrons remain as nonbonded – (number of electrons involved in bond formation/2).The structural representation having maximum number of zero formal charge of its respective atoms will be the most stable lewis structure. The calculation of formal charge of each of the atom in a molecule is very much significant in chemistry because it helps to detect the most stable lewis structure. As there is only one lone pair, lone pair-lone pair repulsion is insignificant for this molecule.įor this bond pair-bond pair repulsion and lone pair – bond pair repulsion, this molecule is deviated from its actual geometrical structure (tetrahedral) and shows a trigonal pyramidal structure with three bond pairs and one lone pair on central atom, arsenic. These two lone electrons face repulsion from the bonding electrons. Rest of the two valance electrons remain as nonbonded. Among these five electrons of arsenic, three electrons participate in covalent bond formation with three fluorine atoms. 
The valance shell electron configuration of As and F are 2s 2 2p 3 and 2s 2 2p 5. In AsF 3, arsenic and fluorine have five and seven electrons in their respective valance shell or outer most shell. The increasing order of the above repulsive factor is-īond pair -bond pair repulsion < Lone pair – bond pair repulsion < Lone pair- lone pair repulsion. Repulsion between bond pair and lone pair can be three types. Repulsion involving lone pair and bond pairs.Geometrical structure only depends upon the hybridization of central atom but shape of any molecule depends upon the following parameters. Shape and structure of any molecule is almost two similar words only if there is no repulsion involving bond pair and lone pair is present. 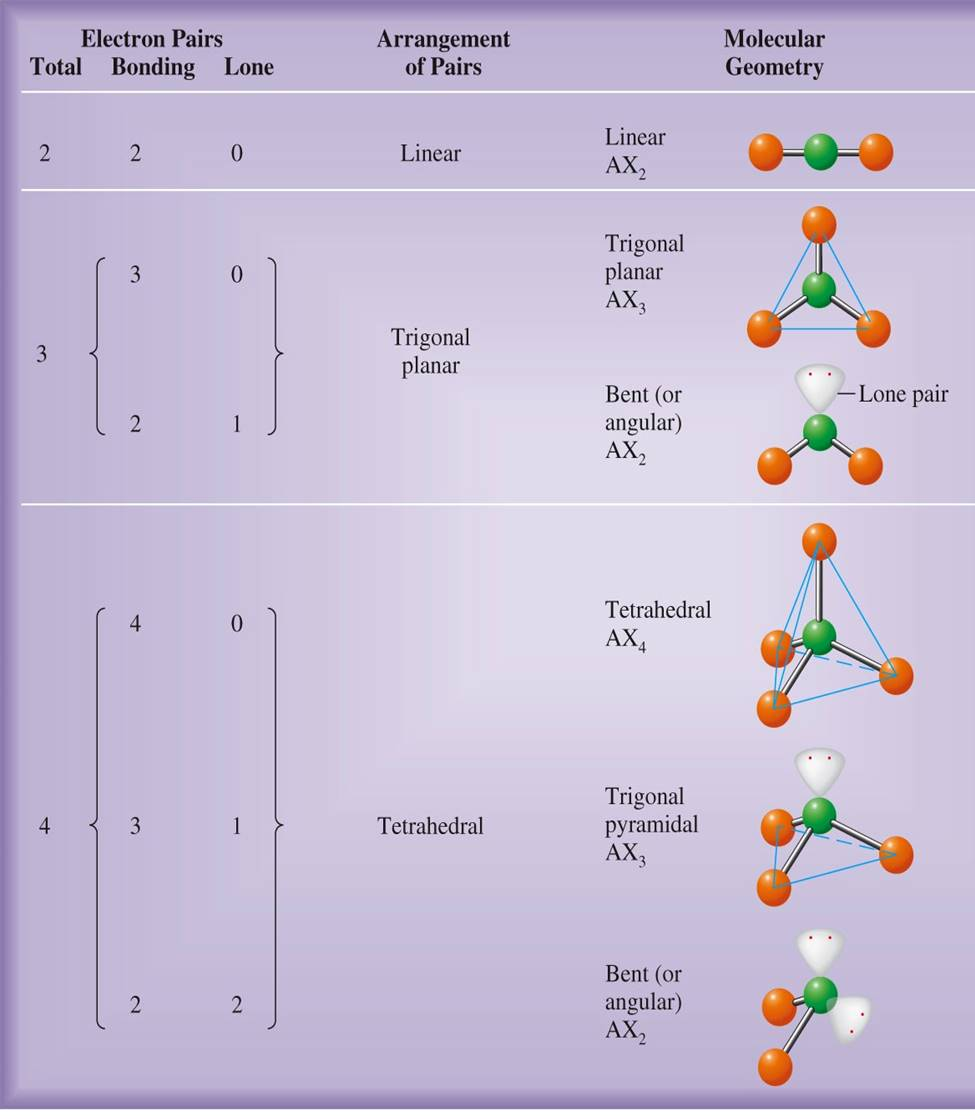
Finding out the nonbonding electrons: Arsenic has two nonbonding electrons and fluorine has total five electrons as nonbonded.ĪsF3 Lewis Structure AsF 3 Lewis Structure Shape.Thus (3×2 = 6) electrons are involved in bonding. Finding out the bonding electrons: Total three covalent bonds are present in this molecular species.Determination of valance electron: In this molecule, AsF 3, central atom, arsenic and substituent atoms fluorine have three and five electrons respectively in their outer most shell.It is basically a structural representation of a molecule where the nonbonding electrons are shown around the respective participating atoms in the lewis structure. To know the process of drawing a lewis structure, first you have to know what is lewis structure. Let’s focus on the following topics on arsenic trifluoride. Arsenic is sp 3 hybridized in AsF 3 molecule in which three bond pairs and one lone pair are present. It is pyramidal structured with bond angle (F-As-F) 96.2 0 and As-F bond length is 170.6 pm. This structure influences its reactivity and use in chemical synthesis.Īrsenic trifluoride is mainly a gaseous compound but it is also found in solid state also. The As-F bonds are polar because of the electronegativity difference (As: 2.18, F: 3.98), contributing to the molecule’s overall polarity. AsF3 exhibits a trigonal pyramidal geometry, with bond angles slightly less than 109.5° due to the lone pair on arsenic, indicating sp³ hybridization. The Lewis structure shows three single As-F bonds and a lone pair on the As atom, using 26 valence electrons. 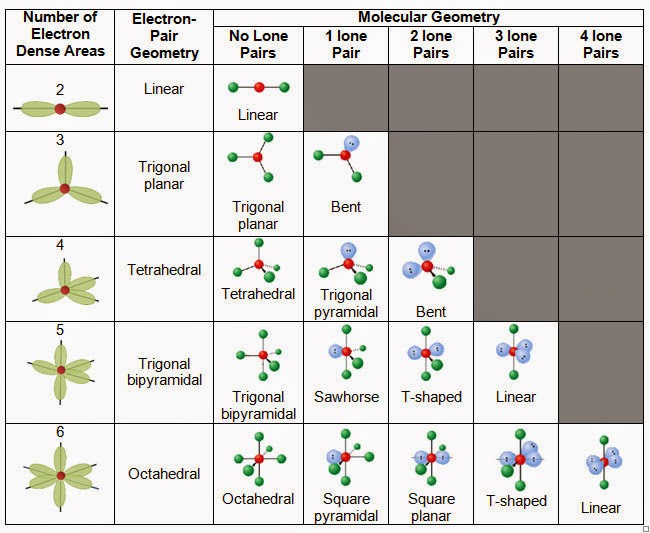
The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible.Arsenic trifluoride (AsF3) has a central arsenic (As) atom with 5 valence electrons, bonded to three fluorine (F) atoms, each contributing 7 valence electrons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
